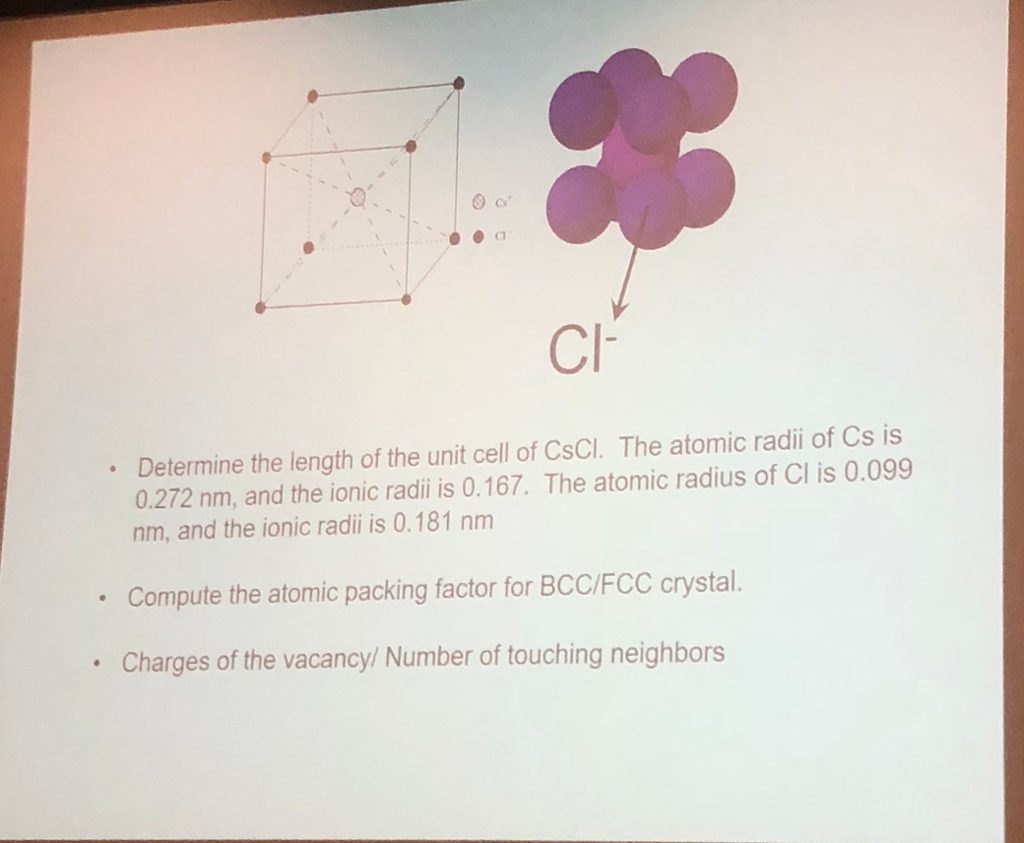
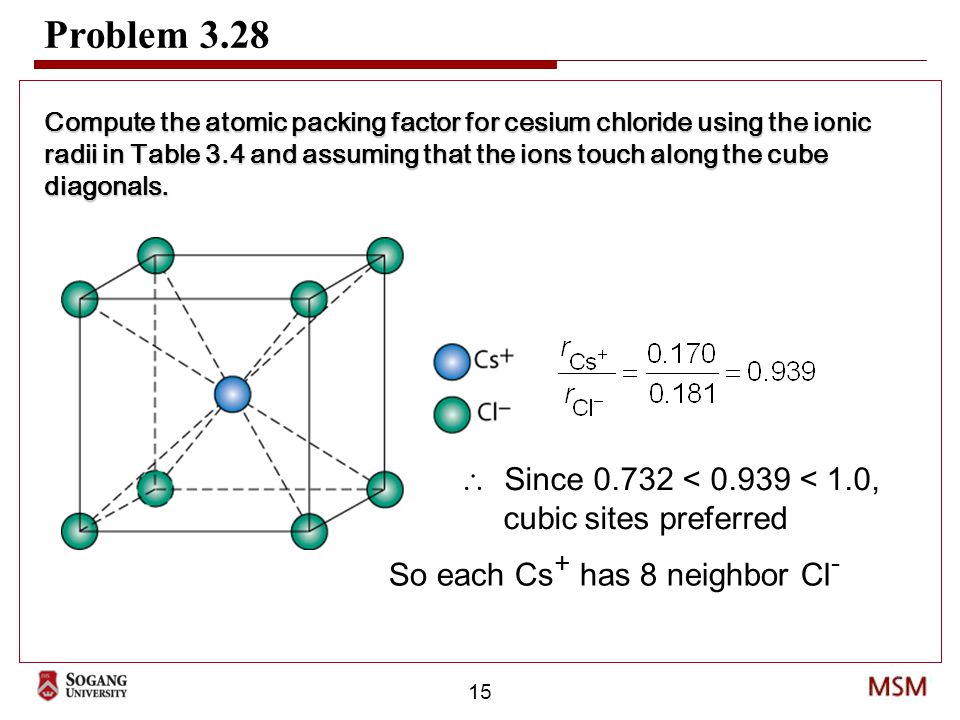
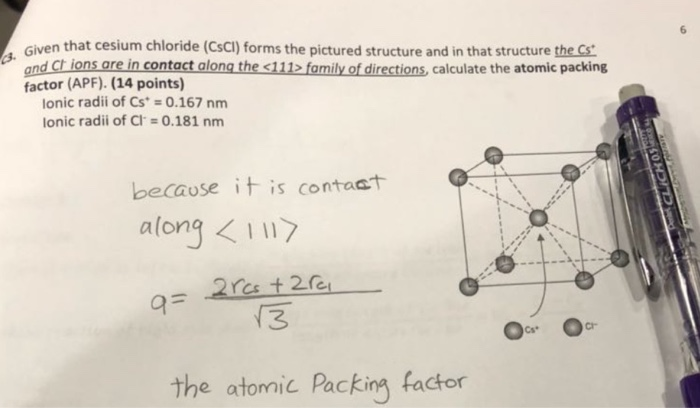
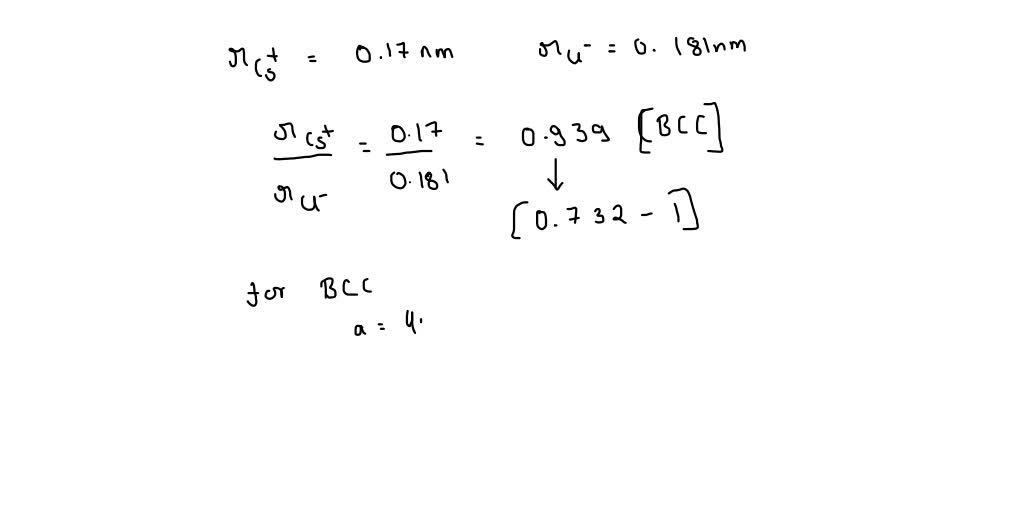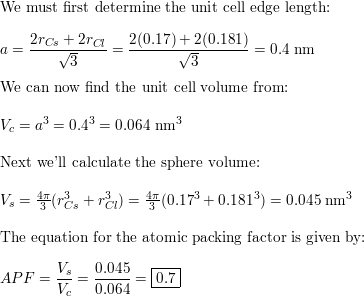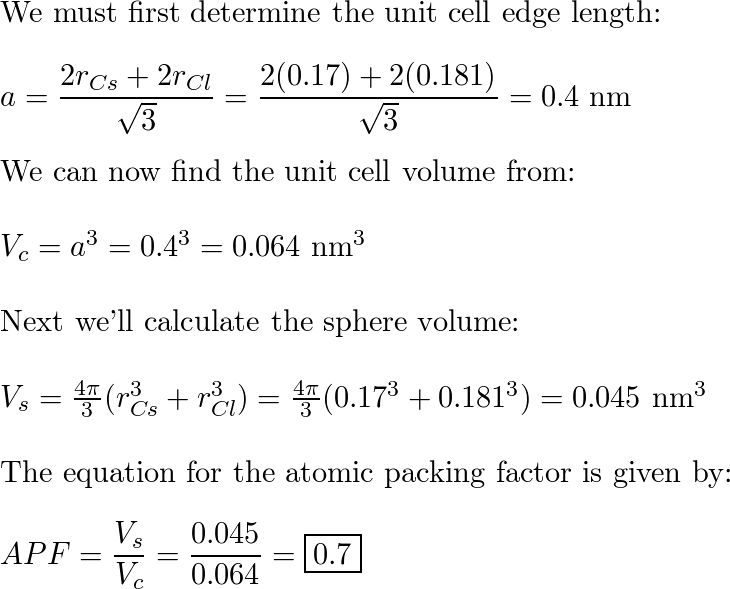CsCl has cubic structure of ions in which `Cs^(+)` ion is present in the body -centre of the cube. - YouTube
Chapter 3: Packing Densities and Coordination Re-cap: Atomic Packing Factor (APF) cap: Atomic Packing Factor (APF)

A metal has bcc structure and the edge length of its unit cell is 3.04 oA . Find the volume of the unit cell in cm^3 .
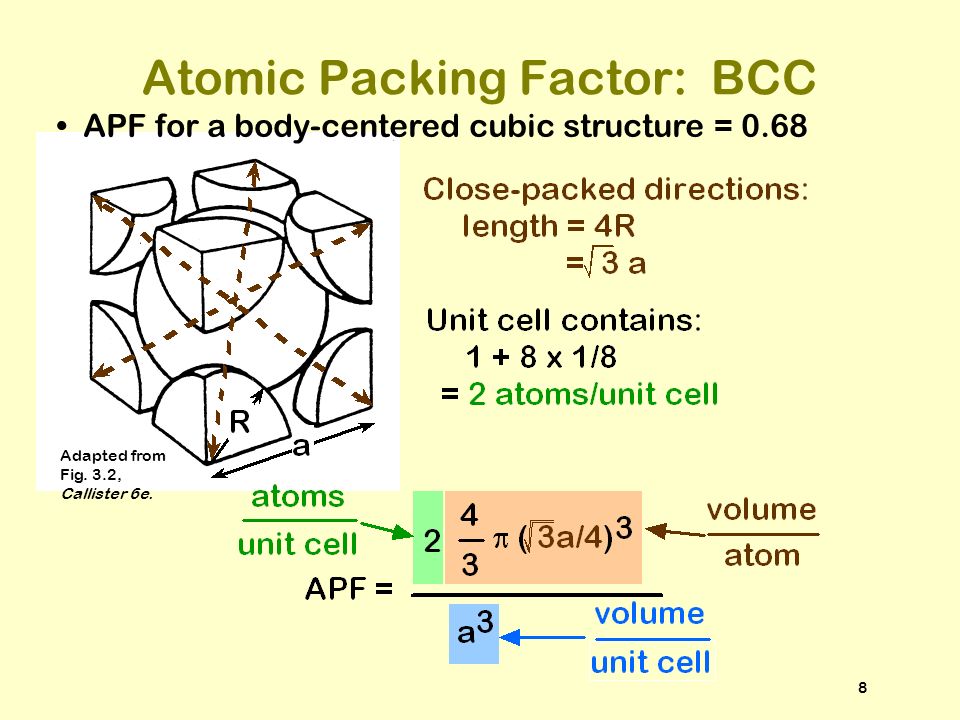
ISSUES TO ADDRESS... How do atoms assemble into solid structures? (for now, focus on metals) How does the density of a material depend on its structure? - ppt download
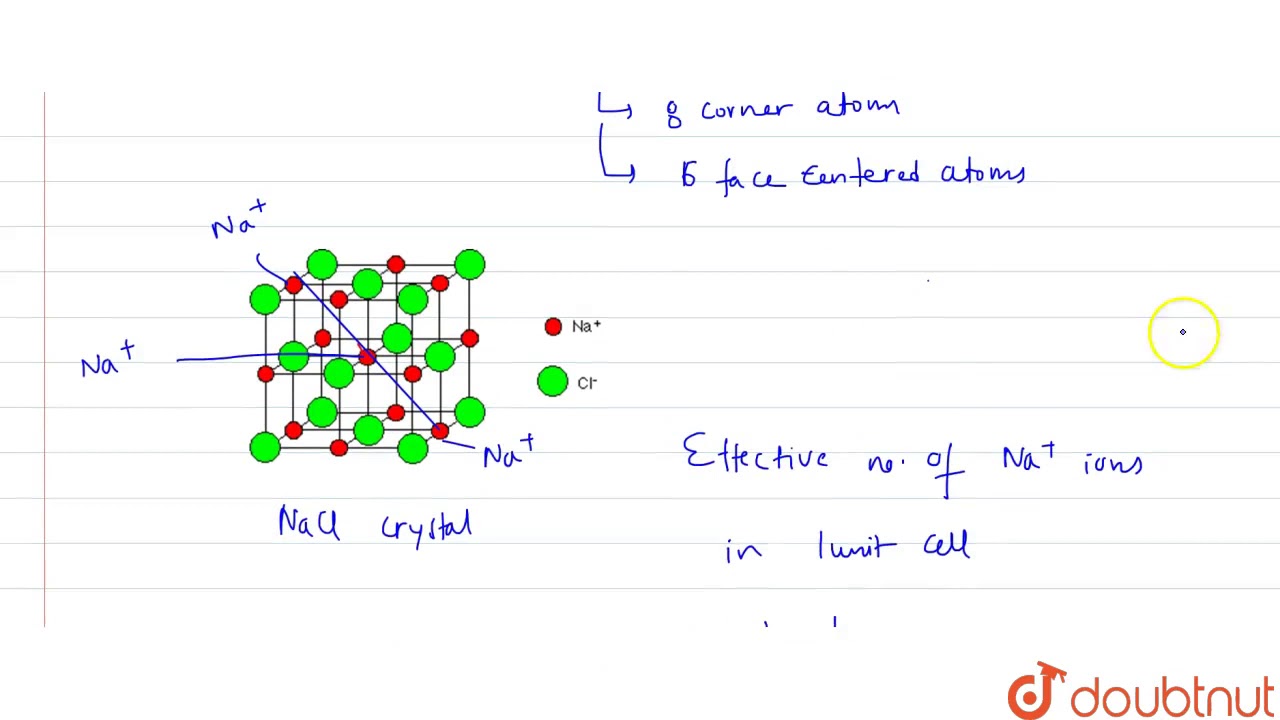
Select right expression for determinig packing fraction (P.F.) of NaCl unit cell (assume ideal )... - YouTube